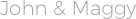- Blog
- Harley davidson golf cart parts for sale
- Tsuki no ookisa full
- Adobe illustrator cc 2017
- Texture pack for minecraft pe
- Darkroom booth manual
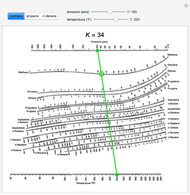
- Depriester chart high temperatures
- Adobe creative suite 5-5 student version
- Mesozoica gameplay
- App icon generator xcode
- Omnifocus for pc
- Reason 6 third party plugins
- How to run a chi square test in spss
- Street love mod gta sa pc
Rectally - Temperatures taken rectally tend to be 0.5 to 0.7 degrees F HIGHER than when taken by mouth.Orally - By mouth - glass thermometer, or a digital thermometer.It is generally medically accepted that normal body temperature ranges between 36.5☌ (97.7☏) to 37.5☌ (99.5☏).A temperature over 38☌ (100.4☏) most often means you have a fever caused by infection or illness.Body temperature may be abnormal due to fever (high temperature) or hypothermia (low temperature).Body temperature can change throughout the day.(1)īody temperature thermometer Quick Facts: It's important to clarify that not all COVID-19 cases will get a fever, nor will everyone with a fever test positive for COVID-19, but it is an extremely common symptom. Or 1.1☌ above the person's usual value.Older adults: 37.8☌ (100☏) and above (oral temperature).Adults: 38☌ (100.4☏) and above (oral temperature).Children: 38☌ (100.4☏) and above (rectal temperature).87.9% of 55,924 laboratory-confirmed cases reported a fever.The armpit is about 0.5C lower than the human body's core temperature (temperature of the internal organs) - the time of day, your age, and activity level, and, in women, the stage of menstrual cycle can also affect your temperature reading.Īccording to the World Health Organization (WHO): OpenPandemics COVID-19 World Community Grid Project.Convert MS Word Docs to Plain Text and HTML.Solutions show the typical behavior of the pure liquid with temperature but the density is also strongly affected by the quantity of dissolved material. Most other pure liquids are like ethanol in this respect. Unlike the situation with water, there is no maximum density point. We can see that the density decreases with temperature through this range. The table at right has the density of ethanol from 3 deg to 40 deg C in g/mL.

It doesn't have the same type of 3-dimensional lattice that water has though. It is similar to water in that it is polar, with a permanent dipole moment, and forms hydrogen bonds with itself. Other Pure Liquids Pure ethanol, CH 3CH 2OH, is another pure liquid. Adding a crystal to supercooled water causes instantaneous ice formation. The long range structure is not perfect but the short range structure of supercooled water is very much like ice.
#DEPRIESTER CHART HIGH TEMPERATURES FREE#
Molecules in this supercooled water are free to move. It is possible to have liquid water at temperatures well below 0 deg C. At low temperature, more of the water has the same lattice as ice. The bonds in water break more slowly as temperature decreases and the structure tend to trap fewer extra water molecules. This is the reason why liquid water is more dense than solid water. Liquid water at 25 deg is so rapidly breaking bonds between H 2O units and reforming them that extra water molecules get trapped inside the water lattice. Remember that liquid water and solid water have the same network of bonds. Density increase as the temperature decreases.īelow 4 deg C, however, the density decreases again. We see the same trend in going from liquid water at 25 deg C (0.9970 g/mL) to liquid water at 4 deg C (0.99997 g/mL).

Together these mean that each H 2O unit in liquid water takes up more space as the temperature increases. This makes sense because, as heat is added to the liquid water, there is greater kinetic energy of the molecules and there are also more vibrations of the water molecules. The density decreases from 0.9970 g/mL to 0.9718 as it is heated. Let's look at the density of water at 25 deg C and compare that to a higher temperature, 80 deg C. Divide by 10 3 to get the density in g/mL. The chart at right give the density in kg/m 3.
Pure Water The density of liquid water is approximately 1.0 g/mL. We know that, for gases, the volume is directly proportional to temperature by the equation PV=nRT. Remember that temperature is related to the average kinetic energy of the atoms or molecules within the substance. Within a particular phase, how does the density depend on temperature? Water has a higher density in the liquid state than the solid, so ice cubes float. Most materials have a lower density of the liquid than the solid but this isn't always true. Gases always have much lower density than the condensed phases. Temperature Effects on Density Temperature Effects on Densityĭensity is the mass of any material per unit volume.